 Failing this, he concluded that the negative charge and the cathode rays were somehow stuck together.Īll previous attempts to bend cathode rays with an electric field had failed, so Thomson devised a new approach in a second pivotal experiment. The purpose was to determine if, by bending the rays with a magnet, Thomson could separate the charge from the rays. First, in a variation of a pivotal 1895 experiment by Jean Perrin, he built a pair of cathode ray tubes ending in a pair of metal cylinders with a slit in them, which were in turn connected to an electrometer. Three of his experiments proved especially conclusive. 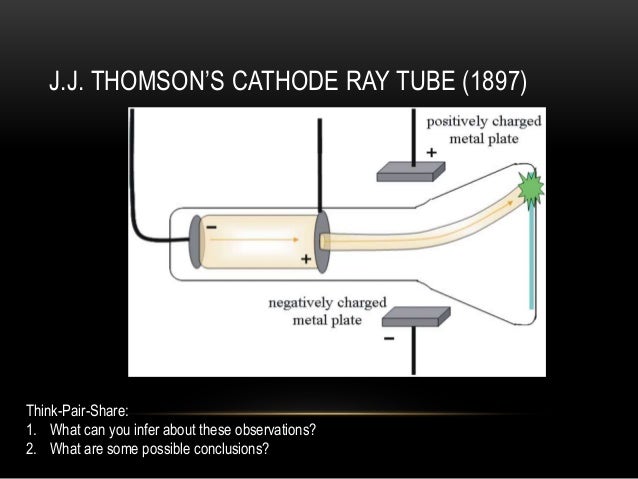
Thomson refined previous experiments and designed new ones in his quest to uncover the true nature of these mysterious cathode rays. Scientists theorized that the glow was produced by some kind of ray emitted by the cathode, but it took the seminal research of a British professor in Cambridge University's Cavendish Laboratory to finally provide a solution to the puzzle. The result: the interior of the tube would glow in lovely fluorescent patterns. They took a glass tube with wires embedded in opposite ends, administered a high voltage and pumped out most of the air. Science lecturers traveling from town to town in the mid-19th century delighted audiences with a device that could be considered the ancestor of the neon sign. Many thanks to MagLab physicist William Coniglio, the science advisor for this page, for his time and expertise.October 1897: The Discovery of the Electron 
The electrons in the cathode rays would deflect toward the positively charged plates, and away from the negatively charged plates. The results showed electrostatic deflection (as opposed to the electromagnetic deflection described above). In addition to experimenting with magnets, scientists also experimented to see what would happen if charged plates were positioned near the tube. (As you apply that rule, remember that the electrons in the cathode ray are travelling opposite the flow of conventional current.) Try flipping the magnet by checking the Flip Magnet box, and observe how the beam then deflects in the opposite direction. That rule describes how a charged particle (our electron) moving in a magnetic field will be deflected by that field at a right angle to both the field and to the direction of the particle. What happens in the tube is a consequence of the Lorentz Force, which is explained by the left hand rule. Thomson would determine that the molecules hypothesized by Crookes were actually negatively charged subatomic particles that he called corpuscles, but which were eventually named electrons. William Crookes experimented with cathode rays and magnets in a similar manner, and his observations on the deflection of the rays by magnetic fields led him to conclude that they were composed of negatively charged molecules. Observe the effect of a magnetic field on cathode rays by using the Magnet Position slider to move a horseshoe magnet (its north pole facing you) so that its poles straddle the cathode ray tube. 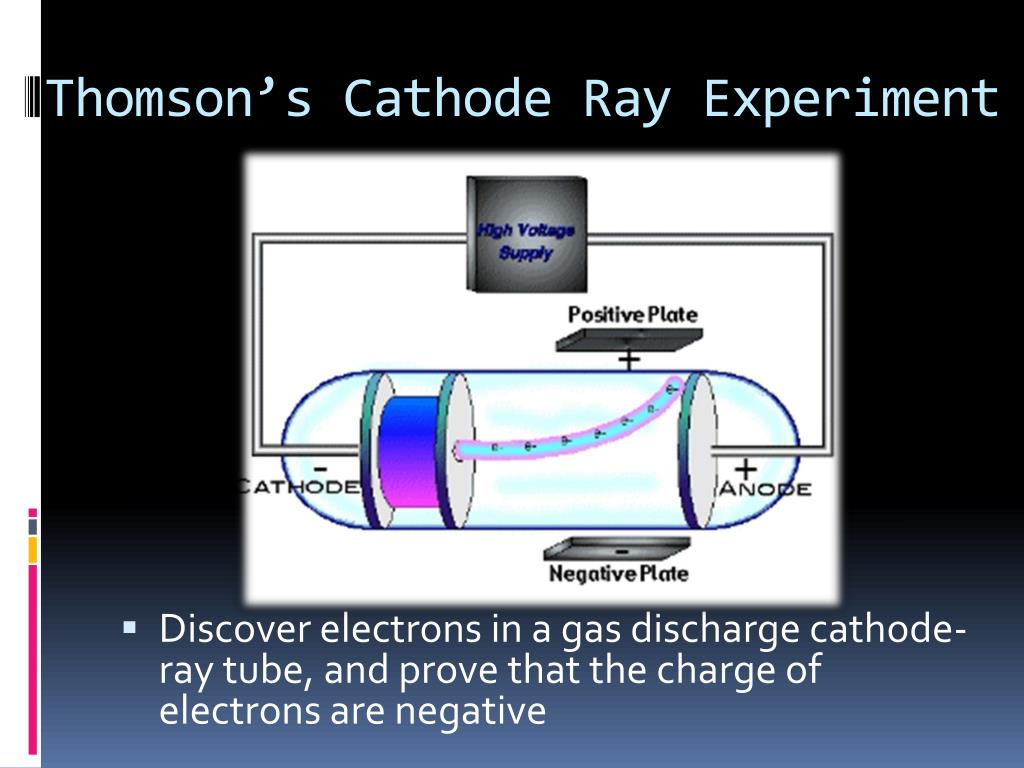
These electrons, or cathode rays, are passed through a small opening near the cathode and then travel in a straight line toward the anode, passing through a fluorescent screen positioned between the cathodes that allows you to see the path of the electrons. A high voltage is transmitted to the cathode ray tube, inducing the cathode to emit electrons – essentially an electrical current. The tube illustrated in the tutorial contains a negative electrode ( Cathode) at one end and a positive electrode ( Anode) at the other. Scientists used special vacuum tubes, such as the Crookes tube and the cathode ray tube, to study this phenomenon. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
